JEE 2026 Chemistry Solving Questions.
Published on Jul 22, 2025 • Education
Introduction
Welcome JEE aspirants!
In today’s chemistry tutorial, we’ll walk through a solved JEE-level chemistry question involving combustion analysis to calculate the percentage composition of hydrogen and oxygen in an organic compound.
This concept is crucial for excelling in:
- JEE Main Chemistry
- JEE Advanced Chemistry
- CBSE Class 11 & 12 Organic Chemistry
- Engineering entrance exams across India
Let’s proceed with a methodical, exam-focused approach to mastering this core topic.
You can watch the full class in the video below:
Question Overview
Problem:
On combustion, 0.98 g of an organic compound (containing carbon, hydrogen, and oxygen) yields 0.27 g of water (H₂O) and 0.7 g of carbon dioxide (CO₂).
You are asked to calculate the percentage composition of hydrogen and oxygen in the compound.
Step 1: Data Extraction
- Mass of compound = 0.98 g
- Mass of water (H₂O) = 0.27 g
- Mass of carbon dioxide (CO₂) = 0.7 g
Step 2: Determine Mass of Hydrogen
Since hydrogen comes from water, we calculate its mass based on the molecular ratio:
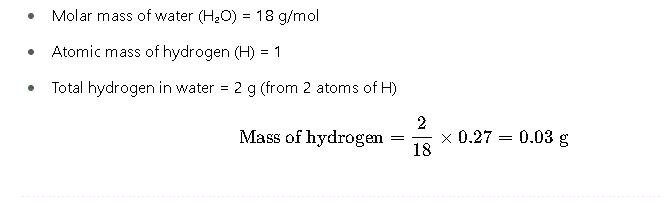
Step 3: Determine Mass of Carbon
Carbon is derived from carbon dioxide (CO₂):
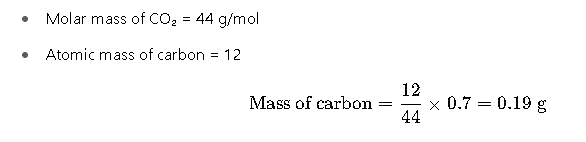
Step 4: Calculate Mass of Oxygen
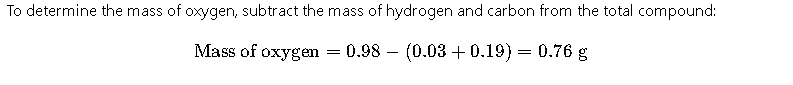
Step 5: Calculate Percentage Composition
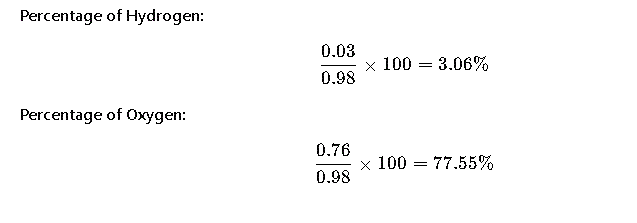
Final Answer
- Hydrogen: 3.06%
- Oxygen: 77.55%
Watch more educational videos and past questions: https://youtube.com/@allcbts
Conclusion
Understanding how to calculate the percentage composition of elements through combustion analysis is a vital skill for success in JEE Main and Advanced Chemistry.
In this tutorial, we:
- Identified the role of water and carbon dioxide in combustion
- Used molar mass ratios to calculate elemental masses
- Applied stoichiometric principles to find percentage composition
- Keep practicing similar questions and focus on concepts like empirical formula, mole calculations, and organic compound identification — all of which are frequently tested in JEE exams.
Related Videos:
- Understanding Waves and Properties in Physics
- Physics 100-Level Past Questions
- GAOKAO Top Mathematics Questions
- Quantitative Reasoning Questions for Basic School
- NCEE Verbal Reasoning for Common Entrance Exam
SEO Keywords for JEE Chemistry
- JEE chemistry percentage composition questions
- combustion analysis JEE main
- hydrogen and oxygen composition in organic compounds
- JEE Advanced chemistry solved examples
- stoichiometry questions for JEE
- organic compound analysis JEE
- JEE chemistry mass-mass relationship
Related News


Latest Posts
CBT Practice App
Download the mobile app to take CBT tests, practice real exam questions, and monitor your progress anytime. You can also use the web version.






